 Purely from the assumption of the fluid being isotropic you have to arrive at the conclusion that stress is linearly proportional to strain with a coefficient we call "mu" (and another proportional to divergence we call "lambda"). The wiki version is fine but there are more thorough versions in textbooks. Look up the derivation of the stress-strain relationship in Navier-Stokes, doesn't matter if it's constant density or not. It's only this thing that falls out of the equations after you make a simplifying assumption of constant density. The property called kinematic viscosity has no real meaning in any flow. Then, wouldn't constant nu a better assumption in this case? 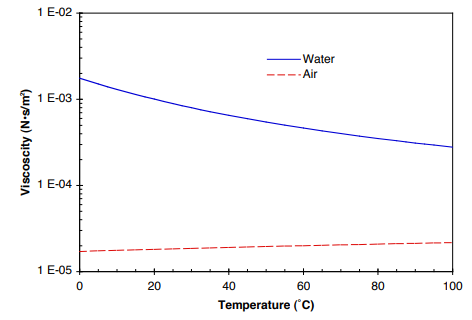
Of course you can have multiphase flow with different properties but you're never going to make any such constant property assumptions there anyway. I specifically was responding to your quoted regime of compressible flow, where yes you get temperature variations with density variations. Your previous reply saying density variation always come with temperature variation using equation of state is not valid when there are multiple phases or components. So it should make some intuitive sense that in a variable density flow, the viscosity coefficient that takes into account density is a more physically meaningful quantity. It should be intuitively true that if you have two substances that are equally "sticky" but one is "heavier", the "heavier" one is going to have a stronger resistance due to inertia. Physically, the viscosity coefficient represents the sort of "strength" of the fluid's resistance to shear. And if density is constant then constant mu also implies constant nu - they're identical statements so your distinction here doesn't make much sense. If the density is variable then mu matters. Who is "people"? If you're dealing with a constant density flow, then nu is the parameter that matters. I am just trying to understand why people favor the constant mu over constant nu assumption How is that relevant to the question? Obviously if you are considering a problem with two totally different fluid properties then assuming constant viscosity or constant density are both very wrong. 
Consider two fluids in a container with low miscibility, the viscosity clearly non-uniform and dependent on local composition or density, while the temperature can be assumed constant I can come up with an example for the case I described. Off hand, you're probably okay with constant viscosity if you're operating in lower mach numbers. If that's "small" then assuming constant viscosity probably won't negatively impact you, particularly for high Reynolds number flows. If you're talking standard air, then look at Sutherland's law and compute the viscosity difference for your expected temperature differences. I'm wondering if these assumptions has any physical basis. I've seem derivations taking mu out of the spatial derivatives in compressible N-S equation. Maybe that's equivalent to the weakly compressible formulations? If you have density variations that's going to need some equation of state and coupling to the first law of thermodynamics, which makes assuming constant temperature a dubious idea. I'm sure someone somewhere formulates their governing equations like this, but it's not common, as far as I'm aware. 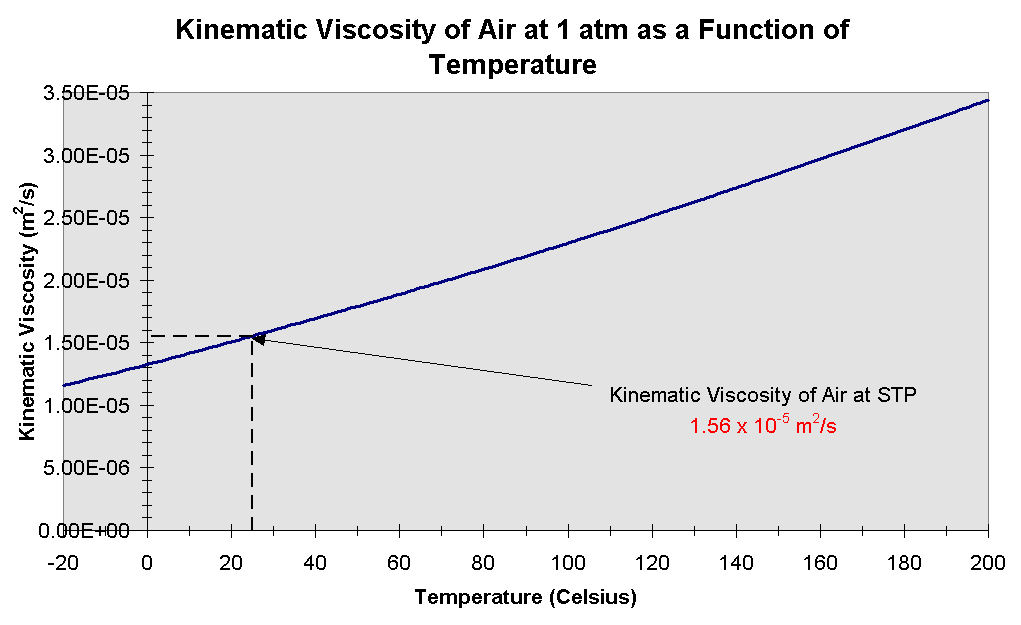
If the temperature is kept constant, but density may vary (compressible) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
